Federal Circuit: Conventional Techniques Do Not Bar Patent Eligibility
Mar 05 2026
In REGENXBIO v. Sarepta Therapeutics, the Federal Circuit recently held that a cell containing a non-naturally occurring recombinant nucleic acid molecule is not a product of nature and is patent‑eligible under 35 U.S.C. § 101, even though the recombinant molecule combines naturally occurring sequences and is incorporated into a cell by conventional technology. While REGENXBIO specifically relates to engineered biological products, it should have broader implications for addressing patent eligibility across all technologies.
The claim at issue of REGENXBIO’s U.S. Patent No. 10,526,617 reads:
It is undisputed by either party, in both the district court and on appeal, that the recombinant nucleic acid molecule, created by joining nucleic acid fragments from two different organisms, does not exist in nature. Likewise both parties agreed that the claimed host cell containing the recombinant nucleic acid molecule does not exist in nature.
In granting summary judgement, the district court found the claims ineligible, reasoning that the claimed compositions merely combine natural products and place them in a host cell without changing any individual component. Analogizing to the ineligible mixed bacterial cultures of Funk Brothers, the court held that combining two sequences from two different organisms is no different than mixing two bacterial strains.
However, the Federal Circuit unanimously reversed, holding that the claims are not directed to a natural phenomenon. The panel emphasized that the claims recite a single recombinant nucleic acid molecule created by joining nucleic acid fragments from different species and introduced into a host cell, thereby “fundamentally creating a cell containing a molecule that could not form in nature on its own” and thus “the claims here are not merely directed to repackaging products of nature.” Having found that the claimed host cells contain molecules markedly different from anything naturally occurring, the court explained it “need not reach the question whether they are markedly different based on unclaimed functions.” Even if considered, Chakrabarty permits looking to “the potential for significant utility,” and the claimed compositions are beneficial for gene delivery in a way their isolated constituents are not. The court also rejected attempts to disregard “conventional” claim elements in the relevant § 101 analysis, and reiterated that the claims must be assessed as a whole rather than by dissected non-conventional components.
Because the claims satisfied step one as not directed to a product of nature, the court did not reach step two of the Alice/Mayo framework.*
* Alice step one – determine whether the claims are directed to a patent-ineligible concept;
Alice step two – determine whether the claim’s elements, considered both individually and as an ordered combination, transform the nature of the claims into a patent-eligible application.
The following table summarizes how the Supreme Court and Federal Circuit have evaluated composition claims under § 101 in representative cases as referenced in the REGENXBIO decision:
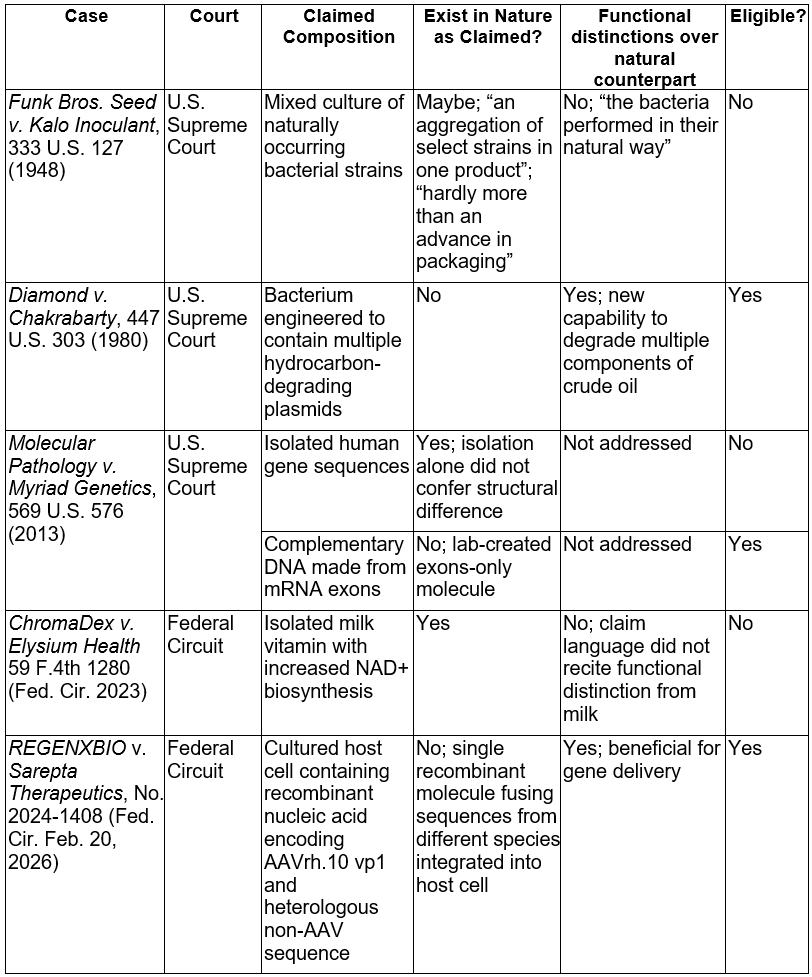
In REGENXBIO, the Federal Circuit clarified patent eligibility of biological products by rejecting the excision of conventional elements at step one of the Alice analysis and requiring a holistic claim analysis. That reasoning may not be limited to biological compositions and should extend to other judicial exceptions, including abstract ideas and laws of nature. In parallel, the USPTO has implemented new tools to address eligibility, including its August 4, 2025 guidance for examiners on evaluating subject matter eligibility of claims, the Director’s In re Desjardins decision on September 26, 2025, and introduction of the term Subject Matter Eligibility Declaration (SMED) in the December 4, 2025 memoranda, all reflecting a broader trend of promoting patent eligibility. Although the Patent Eligibility Restoration Act of 2025 was reintroduced in both chambers on May 1, 2025, its prospects remain uncertain. In the meantime, the USPTO’s evidentiary pathways and the Federal Circuit’s REGENXBIO framework provide concrete administrative and judicial routes to secure protection, especially where claims recite non-natural features or concrete technological improvements.